June 22, 2017

The idea of creating new high-speed mass transit systems is a hot topic for many scientists around the world. Spurred on by technology engineering entrepreneur, Elon Musk, the race is on to create pods that can transport passengers and cargo at high speeds. Musk, the founder and co-founder of several innovative companies including Tesla (premium electric cars), SolarCity (solar energy supply), and PayPal (online payment portal), has inaugurated the Hyperloop project with one of his companies, SpaceX. Although SpaceX was originally set up to explore aerospace manufacture and transport, with the ultimate goal of enabling colonization of other planets, Musk recognized that harnessing the expertise of aeronautical engineers could revolutionize ground transport, and so set up Hyperloop.
The Hyperloop concept is being developed so that pods will travel at very high speeds along a tube, to provide an alternative to flying, rail transportation and driving, while also being safe, convenient and kinder to the environment in not using power that is dependent on fossil fuels. There are several Hyperloop sites being established around the world, and in 2015 Musk launched a competition to encourage the best international student groups to push the boundaries of science and create their ideas of how pods could operate.
Two such top universities, Massachusetts Institute of Technology (MIT, in the US) and Delft University of Technology (TU Delft, in the Netherlands), took part in the first phase of a competition in January this year to unveil their prototype pods at a Hyperloop test track in California. Challenged with a tunnel of one mile in length and six-feet diameter, the teams had to demonstrate that their pods could safely accelerate to speeds of up to 250 mph and break to a standstill without crashing. Both teams used magnetic levitation as their method of propulsion.
For more information, click
here.
June 14, 2017
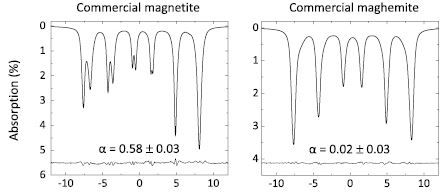 The composition or stoichiometry of magnetite and maghemite mixtures or solid solutions is an important parameter in the physical and geological sciences, and materials science. It is also significant in biomedical science, where magnetic nanoparticles are used both in vitro and clinically, and where both ferrous and ferric iron ions are known to play active roles in the production of reactive oxygen species. However, the accurate determination of the composition/stoichiometry can be problematic, as it requires either well-crystallised samples suitable for x-ray diffraction studies, or it relies on destructive testing methods involving chemical dissolution that, depending on the nature of the sample, are often either unfeasible or inappropriate.
The composition or stoichiometry of magnetite and maghemite mixtures or solid solutions is an important parameter in the physical and geological sciences, and materials science. It is also significant in biomedical science, where magnetic nanoparticles are used both in vitro and clinically, and where both ferrous and ferric iron ions are known to play active roles in the production of reactive oxygen species. However, the accurate determination of the composition/stoichiometry can be problematic, as it requires either well-crystallised samples suitable for x-ray diffraction studies, or it relies on destructive testing methods involving chemical dissolution that, depending on the nature of the sample, are often either unfeasible or inappropriate.
Quentin Pankhurst together with different other groups from Denmark and Spain recently published a thorough paper on the determination of magnetite/maghemite ratios in magnetic nanoparticles. They used Fe-57 Mössbauer spectroscopy and curve-fitted it and then extracted the so-called "centre of gravity" parameter, which is the area weighted mean isomer shift at room temperature.
The authors focused in their paper on the application of the method to materials for which the more conventional techniques are not easily applied—such as in nanoparticles or poorly crystallised particles. It is somewhat salutary to note that the Mössbauer method may find application even in more routine assays.
Check out the paper here.
June 13, 2017
.JPG) The basic idea in 1997, when we started the first ferrofluid workshop in Bremen (at that time „Bremer Ferrofluidworkshop“) had been to coordinate the few groups working with ferrofluids in Germany a bit better and to induce a somewhat stronger interaction. The result of this activity had been the establishment of the first DFG priority program for this field of research – the SPP 1104. In addition a series of conferences has been established – now lasting for 20 years.
The basic idea in 1997, when we started the first ferrofluid workshop in Bremen (at that time „Bremer Ferrofluidworkshop“) had been to coordinate the few groups working with ferrofluids in Germany a bit better and to induce a somewhat stronger interaction. The result of this activity had been the establishment of the first DFG priority program for this field of research – the SPP 1104. In addition a series of conferences has been established – now lasting for 20 years.
The 16th German Ferrofluid Workshop (only 16, not 20 since no workshops had been held in the years of the international ICMF conferences) stands therefore for a remarkable anniversary. 20 years with continuous development of the research field, with the establishment of a second DFG priority program and with permanently growing interest in the national research community are a clear indication that a sustainable structure has been developed.This anniversary will be celebrated this year in Dresden – where the workshop has never been held before. Therefore we invite you for the 16th German Ferrofluid Workshop from July 17 - 19, 2017 in Dresden. The usual details for the conference, the registration and all further details can be found on the website under www.magnetofluiddynamik.de/ffworkshop16th/.
Stefan Odenbach is looking forward to welcome you in Dresden mid of July!
June 13, 2017

Migratory birds make epic journeys of thousands of kilometers each year, navigating with the help of Earth’s magnetic field. Yet the mechanism behind the biological compass that enables them to achieve this astonishing feat remains a mystery. Peter Hore of the University of Oxford is a leading proponent of the idea that this magnetic sense depends on chemical reactions involving excited radicals produced in a protein in the birds’ retinas. Mark Peplow spoke to him about the growing body of evidence behind the hypothesis.
How do we know that birds have a magnetic sense?
Experiments in the 1960s applied magnetic fields to migratory birds in cages. They tried to get out of the cages in different directions, depending on the direction of the field. In controlled experiments, birds can detect the axis of a magnetic field to within 5 degrees, and we suspect it would be more accurate in the wild, under natural conditions.
June 12, 2017
 Uwe Steinhoff from the PTB (Berlin, Germany) has launched a new project, called MagNaStand "Towards an ISO standard for magnetic nanoparticles". This project is a direct spin-off from RADIOMAG activities. There will be a 1st Industrial Stakeholder Workshop taking place at the PTB in Berlin on 4 July 2017. Please have a look at the attached flyer.
Uwe Steinhoff from the PTB (Berlin, Germany) has launched a new project, called MagNaStand "Towards an ISO standard for magnetic nanoparticles". This project is a direct spin-off from RADIOMAG activities. There will be a 1st Industrial Stakeholder Workshop taking place at the PTB in Berlin on 4 July 2017. Please have a look at the attached flyer.
Registration deadline: June 30, 2017
Main objectives:
1. To develop measurement methods and techniques to ensure highly qualified scientific input into the preparation of ISO 19807
2. To summarise metrological knowledge on MNPs gained in this project according to the “Metrological Checklist” ISO/TC 229 N 673, so that further normative documents covering measurement techniques for MNPs (e.g. static magnetic susceptibility, dynamic magnetic susceptibility and specific loss power) can be prepared.
3. To ensure the take up of results from finalised and ongoing FP7 and Horizon 2020 EU research projects on MNPs, e.g., “NanoMag” and “RADIOMAG”, into the international standardisation process.
4. To ensure coordinated participation of European NMIs and stakeholders in ISO’s standardisation process on MNPs to provide highest impact and fastest development of the standard.
5. To facilitate the take up of methods and technology developed in the project by technical committees, e.g., ISO/TC 229 and end-users in biomedicine, mechanical engineering and environmental remediation sectors.
June 12, 2017
 ETH researchers are developing a new and highly effective way of fortifying iron into food and drinks. Protein nanofibrils are formed from native whey protein. The researchers combined these nanofibrils with iron nanoparticles which can be readily absorbed by the body. The iron-coated whey protein nanofibrils can be administered either in powder or liquid form, and the new compound can be easily added to different types of food without affecting their taste or smell or color. Around 1.2 billion people worldwide suffer from iron deficiency, with women worse affected than men. The ingredients are cheap and in plentiful supply. This iron food supplement would also be a good alternative for people living in poor countries who are more prone to iron deficiency than those living in western industrialized nations.
ETH researchers are developing a new and highly effective way of fortifying iron into food and drinks. Protein nanofibrils are formed from native whey protein. The researchers combined these nanofibrils with iron nanoparticles which can be readily absorbed by the body. The iron-coated whey protein nanofibrils can be administered either in powder or liquid form, and the new compound can be easily added to different types of food without affecting their taste or smell or color. Around 1.2 billion people worldwide suffer from iron deficiency, with women worse affected than men. The ingredients are cheap and in plentiful supply. This iron food supplement would also be a good alternative for people living in poor countries who are more prone to iron deficiency than those living in western industrialized nations.
http://swissinnovation.org/news/web/2017/03-170425-a2
June 01, 2017
 Magnetocaloric devices hold the potential to satisfy the rising demand for cooling in the future. One of the challenges remaining is to reduce the high ecological footprint of the permanent magnets driving the magnetic cooling cycle. Existing devices use neodymium-iron-boron (NdFeB)-type permanent magnets, which account for more than 50% of the ecological footprint of the appliance. To overcome this hurdle, TU Darmstadt and Urban Mining Company have built the first working magnetocaloric demonstrator that uses recycled NdFeB as a magnetic field source. Coupling this with the optimization of the magnets and their geometry, it is possible to further reduce the ecological footprint. Together, these two approaches help to position magnetic cooling as a realistic and sustainable cooling technology.
Magnetocaloric devices hold the potential to satisfy the rising demand for cooling in the future. One of the challenges remaining is to reduce the high ecological footprint of the permanent magnets driving the magnetic cooling cycle. Existing devices use neodymium-iron-boron (NdFeB)-type permanent magnets, which account for more than 50% of the ecological footprint of the appliance. To overcome this hurdle, TU Darmstadt and Urban Mining Company have built the first working magnetocaloric demonstrator that uses recycled NdFeB as a magnetic field source. Coupling this with the optimization of the magnets and their geometry, it is possible to further reduce the ecological footprint. Together, these two approaches help to position magnetic cooling as a realistic and sustainable cooling technology.
See more here at TU Darmstadt's website.
March 18, 2017

IBM has created the world’s smallest magnet using a single atom, and stored one bit of data on it. Currently, hard disk drives use about 100,000 atoms to store a single bit. The ability to read and write one bit on one atom creates new possibilities for developing significantly smaller and denser storage devices, that could someday, for example, enable storing the entire iTunes library of 35 million songs on a device the size of a credit card.
Today’s breakthrough builds on 35 years of nanotechnology history at IBM, including the invention of the Nobel prize-winning scanning tunneling microscope. IBM announced it will be building the world’s first commercial quantum computers for business and science. Future scanning tunneling microscope studies will investigate the potential of performing quantum information processing using individual magnetic atoms.
“Magnetic bits lie at the heart of hard-disk drives, tape and next-generation magnetic memory,” said Christopher Lutz, lead nanoscience researcher at IBM Research – Almaden in San Jose, California. “We conducted this research to understand what happens when you shrink technology down to the most fundamental extreme, the atomic scale.”
By starting at the smallest unit of common matter, the atom, scientists demonstrated the reading and writing of a bit of information to the atom by using electrical current. They showed that two magnetic atoms could be written and read independently even when they were separated by just one nanometer, a distance that is only a millionth the width of a pin head. This tight spacing could eventually yield magnetic storage that is 1,000 times denser than today’s hard disk drives and solid state memory chips. Future applications of nanostructures built with control over the position of every atom could allow people and businesses to store 1,000 times more information in the same space, someday making data centers, computers and personal devices radically smaller and more powerful.
The IBM scientists used a scanning tunneling microscope (STM), an IBM invention that won the 1986 Nobel Prize for Physics, to build and measure isolated single-atom bits using the holmium atoms. The custom microscope operates in extreme vacuum conditions to eliminate interference by air molecules and other contamination. The microscope also uses liquid helium for cooling that allows the atoms to retain their magnetic orientations long enough to be written and read reliably.

Beautiful ferrofluid, with a curious and striking 'peak-on-a-peak' effect. Submitted by Quentin Pankhurst.
Search this site with the power of
Last Modified: February 22, 2024 -
Magneticmicrosphere.com © 2024

 The idea of creating new high-speed mass transit systems is a hot topic for many scientists around the world. Spurred on by technology engineering entrepreneur, Elon Musk, the race is on to create pods that can transport passengers and cargo at high speeds. Musk, the founder and co-founder of several innovative companies including Tesla (premium electric cars), SolarCity (solar energy supply), and PayPal (online payment portal), has inaugurated the Hyperloop project with one of his companies, SpaceX. Although SpaceX was originally set up to explore aerospace manufacture and transport, with the ultimate goal of enabling colonization of other planets, Musk recognized that harnessing the expertise of aeronautical engineers could revolutionize ground transport, and so set up Hyperloop.
The idea of creating new high-speed mass transit systems is a hot topic for many scientists around the world. Spurred on by technology engineering entrepreneur, Elon Musk, the race is on to create pods that can transport passengers and cargo at high speeds. Musk, the founder and co-founder of several innovative companies including Tesla (premium electric cars), SolarCity (solar energy supply), and PayPal (online payment portal), has inaugurated the Hyperloop project with one of his companies, SpaceX. Although SpaceX was originally set up to explore aerospace manufacture and transport, with the ultimate goal of enabling colonization of other planets, Musk recognized that harnessing the expertise of aeronautical engineers could revolutionize ground transport, and so set up Hyperloop.
.JPG)
 Migratory birds make epic journeys of thousands of kilometers each year, navigating with the help of Earth’s magnetic field. Yet the mechanism behind the biological compass that enables them to achieve this astonishing feat remains a mystery. Peter Hore of the University of Oxford is a leading proponent of the idea that this magnetic sense depends on chemical reactions involving excited radicals produced in a protein in the birds’ retinas. Mark Peplow spoke to him about the growing body of evidence behind the hypothesis.
Migratory birds make epic journeys of thousands of kilometers each year, navigating with the help of Earth’s magnetic field. Yet the mechanism behind the biological compass that enables them to achieve this astonishing feat remains a mystery. Peter Hore of the University of Oxford is a leading proponent of the idea that this magnetic sense depends on chemical reactions involving excited radicals produced in a protein in the birds’ retinas. Mark Peplow spoke to him about the growing body of evidence behind the hypothesis. Uwe Steinhoff from the PTB (Berlin, Germany) has launched a new project, called MagNaStand "Towards an ISO standard for magnetic nanoparticles". This project is a direct spin-off from RADIOMAG activities. There will be a 1st Industrial Stakeholder Workshop taking place at the PTB in Berlin on 4 July 2017. Please have a look at the attached flyer.
Uwe Steinhoff from the PTB (Berlin, Germany) has launched a new project, called MagNaStand "Towards an ISO standard for magnetic nanoparticles". This project is a direct spin-off from RADIOMAG activities. There will be a 1st Industrial Stakeholder Workshop taking place at the PTB in Berlin on 4 July 2017. Please have a look at the attached flyer. 
 Magnetocaloric devices hold the potential to satisfy the rising demand for cooling in the future. One of the challenges remaining is to reduce the high ecological footprint of the permanent magnets driving the magnetic cooling cycle. Existing devices use neodymium-iron-boron (NdFeB)-type permanent magnets, which account for more than 50% of the ecological footprint of the appliance. To overcome this hurdle, TU Darmstadt and Urban Mining Company have built the first working magnetocaloric demonstrator that uses recycled NdFeB as a magnetic field source. Coupling this with the optimization of the magnets and their geometry, it is possible to further reduce the ecological footprint. Together, these two approaches help to position magnetic cooling as a realistic and sustainable cooling technology.
Magnetocaloric devices hold the potential to satisfy the rising demand for cooling in the future. One of the challenges remaining is to reduce the high ecological footprint of the permanent magnets driving the magnetic cooling cycle. Existing devices use neodymium-iron-boron (NdFeB)-type permanent magnets, which account for more than 50% of the ecological footprint of the appliance. To overcome this hurdle, TU Darmstadt and Urban Mining Company have built the first working magnetocaloric demonstrator that uses recycled NdFeB as a magnetic field source. Coupling this with the optimization of the magnets and their geometry, it is possible to further reduce the ecological footprint. Together, these two approaches help to position magnetic cooling as a realistic and sustainable cooling technology. IBM has created the world’s smallest magnet using a single atom, and stored one bit of data on it. Currently, hard disk drives use about 100,000 atoms to store a single bit. The ability to read and write one bit on one atom creates new possibilities for developing significantly smaller and denser storage devices, that could someday, for example, enable storing the entire iTunes library of 35 million songs on a device the size of a credit card.
IBM has created the world’s smallest magnet using a single atom, and stored one bit of data on it. Currently, hard disk drives use about 100,000 atoms to store a single bit. The ability to read and write one bit on one atom creates new possibilities for developing significantly smaller and denser storage devices, that could someday, for example, enable storing the entire iTunes library of 35 million songs on a device the size of a credit card.
