
Imaging Method Captures Structural Details of Nanoparticles in Liquids
July 24, 2015
 A. Paul Alivisatos and Alex Zettl of the University of California, Berkeley; Jungwon Park and David A. Weitz of Harvard University; and coworkers produced 3D images of angstrom-sized metal particles in solution at near-atomic resolution (Science 2015, DOI: 10.1126/science.aab1343).
A. Paul Alivisatos and Alex Zettl of the University of California, Berkeley; Jungwon Park and David A. Weitz of Harvard University; and coworkers produced 3D images of angstrom-sized metal particles in solution at near-atomic resolution (Science 2015, DOI: 10.1126/science.aab1343).
The imaging technique used by the researchers, based on transmission electron microscopy (TEM), may enable scientists to monitor dynamics of individual particles within a colloid in their native state. Structural details gleaned from the imaging method may also lead to new uses for nanoparticles in catalysis, biological imaging, and other areas.
Drawing on a liquid-sampling technique the researchers reported in 2012 , they packaged a few droplets of colloidal platinum nanoparticles in a nanosized graphene bubble. Then they used a microscope equipped with a highly sensitive detector to zoom in on individual particles and record many short-exposure images.
Because the particles were constantly moving, the images were inherently low quality. But by applying a computational technique the researchers developed for this purpose, they were able to produce high-resolution 3-D reconstructions of randomly moving nonidentical particles.
This work is likely to make an impact in biological imaging, catalysis, and other fields, says Joerg R. Jinschek, a TEM specialist for microscope maker FEI who is based in the Netherlands. Jinschek explains that attaching a colloidal particle to a rigid surface—as has been done for imaging purposes in the past—can alter its structure. Because structure and other properties often depend on a particle’s state or environment, researchers want detailed information about nanoparticles in their natural states, Jinschek says. He adds that being able to image freely moving nanoparticles in liquids, as was done in the new study, will help scientists determine relationships between a nanoparticle’s structure and its function.
Report from the Frontiers in Biomagnetic Nanoparticles Meeting in Telluride 2015
July 22, 2015Jennifer Andrew, Jeffrey Anker and Thompson Mefford organized now already for the 4th time the Frontiers in Biomagnetic Nanoparticles meeting. It took place in Telluride, Montana, U.S.A. from June 23-25, 2015, a wonderful location high in the Rockies (2590 m above sea level!). With 80 participants, this meeting is smaller than its "twin meeting", the International Conference on the Scientific and Clinical Applications of Magnetic Carriers, but it features just as high-standing talks in the (odd) years between the larger conference.
This time, we had an especially interesting time during discussions about how magnetic hyperthermia works, why it seems to work even without (measurable) heat production, and if we actually should rename the effect to thermophoresis. Especially Tim St. Pierre, his student Michael McPhail and Julian Carrey brought up many interesting points, and we are sure that the discussions will go on during next year's Magnetic Carrier meeting.
Another interesting area that quite a few talks were discussing was the use of magnetic nanoparticles as T2 (and T1) contrast agents in Magnetic Resonance Imaging. Especially the group from NIST Boulder gave some interesting talks, while the shape anisotropy was also mentioned as very important. Jin Xie very nicely reviewed the area and their recent developments and showed how Fe5C2 nanoparticles might replace some of the Gd-based agents in the future.

The next Frontiers of Biomagnetic Particles meeting will take place in 2017 again. Make sure you write it into your calendar!
Successful Conference in Timisoara - June 25-26 , 2015
July 17, 2015
 The Magnetic Fluids & Applications meetings are organized in Timisoara since 1980 by the Politehnica University of Timisoara and the Timisoara Branch of the Romanian Academy. The workshop Multifunctional nanoparticles, magnetically controllable fluids,
The Magnetic Fluids & Applications meetings are organized in Timisoara since 1980 by the Politehnica University of Timisoara and the Timisoara Branch of the Romanian Academy. The workshop Multifunctional nanoparticles, magnetically controllable fluids,  complex flows, engineering and biomedical applications held in the period 25-26 June, 2015 with 16 oral presentations, among them 3 keynote lectures and 5 invited talks, proved to be an excellent opportunity for scientists, representatives of industry, master and PhD students from Germany, Russia, Slovakia, Hungary and the host country Romania to have very stimulating discussions towards cooperation for scientific and technological progress in the field.
complex flows, engineering and biomedical applications held in the period 25-26 June, 2015 with 16 oral presentations, among them 3 keynote lectures and 5 invited talks, proved to be an excellent opportunity for scientists, representatives of industry, master and PhD students from Germany, Russia, Slovakia, Hungary and the host country Romania to have very stimulating discussions towards cooperation for scientific and technological progress in the field.
The abstracts of oral and poster presentations may be seen here.
Latest Status on MPI Research
July 15, 2015
 MPI is a tomographic imaging technique that detects the magnetic properties of iron-oxide nanoparticles injected into the bloodstream to produce 3D images. The new technology, invented by Philips, and first introduced in a 2005 Nature paper, gave rise to a new era in biological imaging. The first commercial machines to do Magnetic Particle Imaging are now available from Bruker. Check out their assessment of the technology (and advertisement) on their website:
MPI is a tomographic imaging technique that detects the magnetic properties of iron-oxide nanoparticles injected into the bloodstream to produce 3D images. The new technology, invented by Philips, and first introduced in a 2005 Nature paper, gave rise to a new era in biological imaging. The first commercial machines to do Magnetic Particle Imaging are now available from Bruker. Check out their assessment of the technology (and advertisement) on their website:
https://www.bruker.com/industries/preclinical-imaging/mpi.html
The most recent international meeting about MPI was in Istanbul from March 26-28, 2015. And the next meeting will be in Lübeck, Germany, from March 16-18, 2016. Check out details about earlier and upcoming MPI meetings here:
Magnetic Particle Imaging Webinar Series
June 03, 2015
 Magnetic Insight is proud to launch the first webinar series on Magnetic Particle Imaging, a unique, ultra-sensitive, high resolution molecular imaging approach that longitudinally detects nanoparticles regardless of depth. Sign up for all three!
Magnetic Insight is proud to launch the first webinar series on Magnetic Particle Imaging, a unique, ultra-sensitive, high resolution molecular imaging approach that longitudinally detects nanoparticles regardless of depth. Sign up for all three!
Tuesday June 9, 2015 9:00 PST/ 18:00 CET The Basics of MPI
Tuesday July 7, 2015 9:00 PST/ 18:00 CET Preclinical Applications in MPI
Tuesday August 4, 2015 9:00 PST/ 18:00 CET Nanoparticle Development for MPI Applications
World Record Speed of a Maglev Train: 603 km/h
April 21, 2015
 The Central Japan Railway company reports that its magnetic levitation bullet train topped 603 km/h on Tuesday during a test run along a length of test track in the Yamanashi prefecture. This was enough to break its own 12-year-old, 361 mph world record set back in 2003. The train reportedly carried 29 engineers during its run.
The Central Japan Railway company reports that its magnetic levitation bullet train topped 603 km/h on Tuesday during a test run along a length of test track in the Yamanashi prefecture. This was enough to break its own 12-year-old, 361 mph world record set back in 2003. The train reportedly carried 29 engineers during its run.
Unfortunate is the fact that normal passengers will likely never be able to experience these exhilarating speeds -- unless something goes horribly wrong. The rail company plans to limit the trains to a pokey 313 mph for regular service when they come online in 2027. But even at these speeds, commuters could make it from Tokyo to Nagoya in about 40 minutes (less than half the time today's fastest bullet trains require). The company even has aspirations to export this technology to America -- specifically as a high-speed rail line running between New York City and Washington DC.
Use of Moessbauer Spectroscopy for Careful Assessment of Magnetic Nanoparticles
March 26, 2015
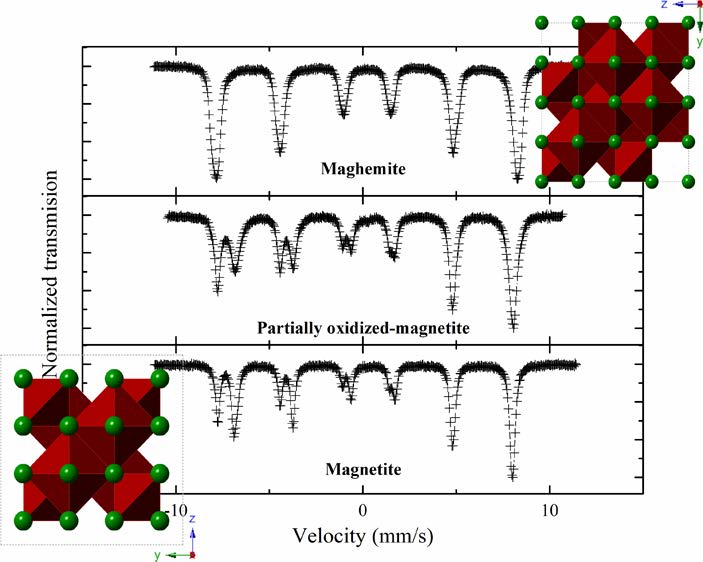 The use of Mossbauer spectroscopy is a simple and direct way to determine the amounts of magnetite and maghemite in an otherwise unknown sample. Quentin Pankhurst et al wrote a nice paper about how to use it and determine magnetite/maghemite weighting directly from the mean isomer shift of the total spectrum, which means that one can apply a simple model-independent curve-fitting methodology. Compared to the older way of using Moessbauer, this takes an awful lot of the work out of the process.
The use of Mossbauer spectroscopy is a simple and direct way to determine the amounts of magnetite and maghemite in an otherwise unknown sample. Quentin Pankhurst et al wrote a nice paper about how to use it and determine magnetite/maghemite weighting directly from the mean isomer shift of the total spectrum, which means that one can apply a simple model-independent curve-fitting methodology. Compared to the older way of using Moessbauer, this takes an awful lot of the work out of the process.
Check out the paper for yourself here.
How to Reliably Measure the SAR of Magnetic Hyperthermia Materials
March 26, 2015
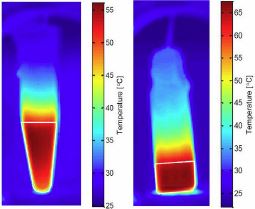
Check out this very useful paper here.
For more information, check out our Archives.
September 2017

Search this site with the power of
