
Magnetic Carrier Meeting 2014
February 15, 2015
 Our already 10th International Conference on the Scientific and Clinical Applications of Magnetic Carriers took place in Dresden, Germany from June 10-14, 2014. It was a wonderful gathering of more than 330 participants from 41 countries, where we discussed all the different aspects of magnetic nanoparticles and microspheres, how they can be made, used and applied in new and ever more fascinating ways!
Our already 10th International Conference on the Scientific and Clinical Applications of Magnetic Carriers took place in Dresden, Germany from June 10-14, 2014. It was a wonderful gathering of more than 330 participants from 41 countries, where we discussed all the different aspects of magnetic nanoparticles and microspheres, how they can be made, used and applied in new and ever more fascinating ways!
Check out all the details at
http://magneticmicrosphere.com/meeting-tenth
The research discussed at the meeting has now been published in full-size peer-reviewed papers in the Journal of Magnetism and Magnetic Materials (JMMM). Check it out here.
How Good Are We at Making Magnetic Nanoparticles?
January 10, 2015

Many parameters of the synthesis procedure may have a strong effect on the particles obtained, including temperature, reagent concentrations, surfactant concentrations, and stirring conditions. This is one of the reasons why scaling-up of some of these synthesis routes is extremely complicated. Indeed, one of the difficulties that particle synthesis faces is in batch-to batch reproducibility. This has led to recent work on alternative reaction platforms that can offer more consistent results. One such platform is the use of microwave irradiation as a heating source.
Check out this interesting paper here.
Correction to "Synthesis of Magnetic Noble Metal (Nano)Particles" Paper
January 03, 2015

spectroscopy (EDS) mapping. Thank you for being honest and pointing our magnetic particle community to possible problems with today's highly sensitive analysis methods.
Inorganic Janus Particles for Biomedical Applications
January 03, 2015

Check this excellent review out here.
Stabilization of the Surfaces of Magnetite
December 20, 2014
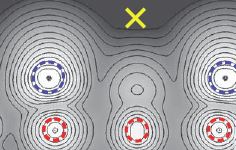
Check it out at Science 5 December 2014:Vol. 346 no. 6214 pp. 1215-1218, DOI:10.1126/science.1260556
Handling of Magnetic Beads in Microfluidics
December 15, 2014

To read more, check out Spinomix website.
Smart Application of Magnetic Fields Allows for Deep Central Focusing of Magnetic Nanoparticles
December 15, 2014
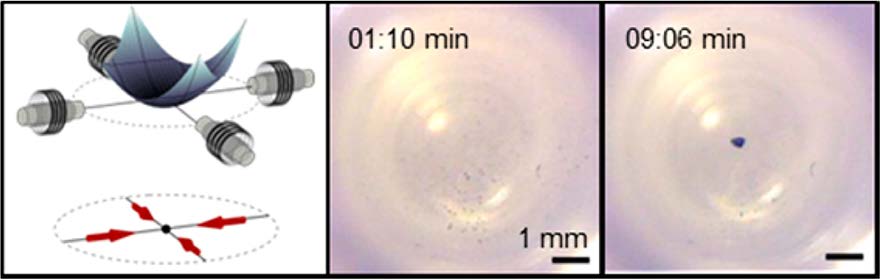 Recent efforts between the University of Maryland (UMD) and Bethesda-based Weinberg Medical Physics LLC (WMP) have led to a new technique to magnetically deliver drug carrying particles to hard-to-reach targets. The method has the potential to transform the way deep-tissue tumors and other diseases are treated. UMD Fischell Department of Bioengineering (BioE) alumnus Dr. Aleksandar Nacev and BioE and Institute for Systems Research Professor Benjamin Shapiro have teamed up with WMP to exploit fast pulsed magnetic fields to focus nano-therapeutic magnetic particles to deep targets.
Recent efforts between the University of Maryland (UMD) and Bethesda-based Weinberg Medical Physics LLC (WMP) have led to a new technique to magnetically deliver drug carrying particles to hard-to-reach targets. The method has the potential to transform the way deep-tissue tumors and other diseases are treated. UMD Fischell Department of Bioengineering (BioE) alumnus Dr. Aleksandar Nacev and BioE and Institute for Systems Research Professor Benjamin Shapiro have teamed up with WMP to exploit fast pulsed magnetic fields to focus nano-therapeutic magnetic particles to deep targets.
Pulsed magnetic fields allowed the team to reverse the usual behavior of magnetic nano-particles. Instead of a magnet attracting the particles, they showed that an initial magnetic pulse can orient the rod shaped particles without pulling them, and then a subsequent pulse can push the particles before the particles can reorient. By repeating the pulses in sequence, the particles were focused to locations deep between the electromagnets.
To find out the details for yourself, check out the Nano Letters paper which is available online at http://dx.doi.org/10.1021/nl503654t with a video showing the magnetic focusing at http://ter.ps/magnetic.
Webinars by Malvern
December 02, 2014
For examples, recent titles are:
The roles of nanoparticles in therapeutic protein aggregation pathways
Powerful protein SEC made simple!
Speed your way through your protein formulation screening by automating your measurements
For more information, check out our Archives.
September 2017

Search this site with the power of
