August 17, 2020
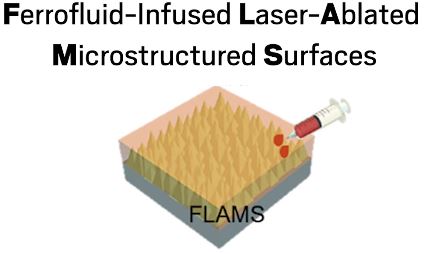 Moving gas bubbles underwater on a surface could help scientists and engineers in the lab and in industry. But it’s not an easy task. Previous methods have relied on slippery silicone oil coatings, but bubbles move slowly on these surfaces and only in one direction. Now, researchers at the University of Science and Technology of China and the Chinese Academy of Sciences have developed a ferrofluid-based surface that lets them move bubbles quickly and in any direction with the help of a magnet (Nano Lett. 2020, DOI: 10.1021/acs.nanolett.0c02091). Using this so-called FLAM surface, the researchers were able to drag bubbles down tilted surfaces and even tow objects more than 500 times their own mass. The authors envision their method being useful for applications such as electrochemical reactions and wastewater treatment.
Moving gas bubbles underwater on a surface could help scientists and engineers in the lab and in industry. But it’s not an easy task. Previous methods have relied on slippery silicone oil coatings, but bubbles move slowly on these surfaces and only in one direction. Now, researchers at the University of Science and Technology of China and the Chinese Academy of Sciences have developed a ferrofluid-based surface that lets them move bubbles quickly and in any direction with the help of a magnet (Nano Lett. 2020, DOI: 10.1021/acs.nanolett.0c02091). Using this so-called FLAM surface, the researchers were able to drag bubbles down tilted surfaces and even tow objects more than 500 times their own mass. The authors envision their method being useful for applications such as electrochemical reactions and wastewater treatment.
August 15, 2020
 Researchers at Empa, the Swiss Federal Laboratories for Materials Science and Technology near Zurich have developed an innovative method to precisely produce different alloy compositions in a single component, using a 3D laser printer. Until recently, 3D printing has presented unique challenges to metal processing, as temperatures nearly instantaneously exceed 2,500 degrees Celsius in the melting zone, which can alter the properties of alloys as some components evaporate. However, a team at Empa, led by Aryan Arabi-Hashemi and Christian Leinenbach, discovered that by varying the power and duration of the laser beam, they could modify the alloys with micrometer precision, thereby “producing new materials with completely new functionalities.” Prospective applications include manufacturing “smart” shape memory alloys and designing more efficient electric motors.
Researchers at Empa, the Swiss Federal Laboratories for Materials Science and Technology near Zurich have developed an innovative method to precisely produce different alloy compositions in a single component, using a 3D laser printer. Until recently, 3D printing has presented unique challenges to metal processing, as temperatures nearly instantaneously exceed 2,500 degrees Celsius in the melting zone, which can alter the properties of alloys as some components evaporate. However, a team at Empa, led by Aryan Arabi-Hashemi and Christian Leinenbach, discovered that by varying the power and duration of the laser beam, they could modify the alloys with micrometer precision, thereby “producing new materials with completely new functionalities.” Prospective applications include manufacturing “smart” shape memory alloys and designing more efficient electric motors.
The picture shows iron filings sticking to a mini chessboard with four millimeter edge length. The partially magnetic structure was produces from a single type of steel power at different temperatures. For more information, check it out here.
August 14, 2020
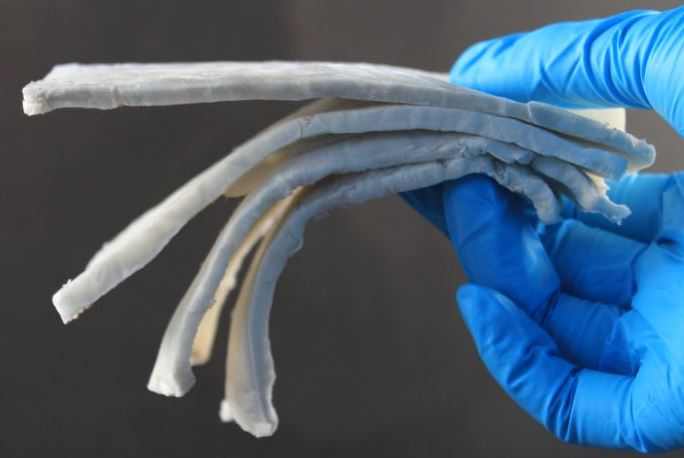 Electric motors and electronic devices generate electromagnetic fields that sometimes have to be shielded in order not to affect neighboring components or the transmission of signals. However, the thin metal sheets or metallized foils that are conventionally used for this purpose are often too heavy or too rigid for many applications. To address this shortcoming, Empa researchers recently succeeded in applying very light aerogels, based on cellulose nanofibers, to microelectronics, which can effectively shield electromagnetic radiation over a wide frequency range. Cellulose fibers are obtained from wood, and due to their chemical structure, enable a wide range of chemical modifications, which are maintained even after being subjected to strong mechanical stress. Combined with two-dimensional nanoplates of titanium carbide, the material has proven to be the lightest electromagnetic shield in the world.
Electric motors and electronic devices generate electromagnetic fields that sometimes have to be shielded in order not to affect neighboring components or the transmission of signals. However, the thin metal sheets or metallized foils that are conventionally used for this purpose are often too heavy or too rigid for many applications. To address this shortcoming, Empa researchers recently succeeded in applying very light aerogels, based on cellulose nanofibers, to microelectronics, which can effectively shield electromagnetic radiation over a wide frequency range. Cellulose fibers are obtained from wood, and due to their chemical structure, enable a wide range of chemical modifications, which are maintained even after being subjected to strong mechanical stress. Combined with two-dimensional nanoplates of titanium carbide, the material has proven to be the lightest electromagnetic shield in the world.
For more details, see here.
August 02, 2020
.JPG) Sampling diseased tissue, which can contain many different types of cells, calls for precise biomedical tools. Current methods for manipulating single cells, like laser microdissection, require complicated setups and can damage cells. Now, researchers led by David H. Gracias at Johns Hopkins University have developed devices that could offer a simpler and less-harmful way to isolate single cells (Nano Lett. 2020, DOI: 10.1021/acs.nanolett.0c01729). Remotely guided by a magnetic field, these so-called microgrippers can wrest single cells from a tissue sample and carry them on demand. Though the tiny grippers require more optimization and testing, the researchers hope one day to use them to perform single-cell biopsies inside a human body.
Sampling diseased tissue, which can contain many different types of cells, calls for precise biomedical tools. Current methods for manipulating single cells, like laser microdissection, require complicated setups and can damage cells. Now, researchers led by David H. Gracias at Johns Hopkins University have developed devices that could offer a simpler and less-harmful way to isolate single cells (Nano Lett. 2020, DOI: 10.1021/acs.nanolett.0c01729). Remotely guided by a magnetic field, these so-called microgrippers can wrest single cells from a tissue sample and carry them on demand. Though the tiny grippers require more optimization and testing, the researchers hope one day to use them to perform single-cell biopsies inside a human body.
For more information, and a movie, check it out here.
June 02, 2020
 Our colleague Dr. Marie Frenea-Robin is the guest editor of a special issue of magnetochemistry, titled "Magnetic Cell Separation". Magnetic cell separation has become a key methodology for the isolation of target cell populations from biological suspensions, covering a wide spectrum of applications from diagnosis and therapy in biomedicine to environmental applications or fundamental research in biology. This Special Issue aims to create a forum of discussion to share advances and address current challenges in magnetic cell separation. For more details, check out this flyer.
Our colleague Dr. Marie Frenea-Robin is the guest editor of a special issue of magnetochemistry, titled "Magnetic Cell Separation". Magnetic cell separation has become a key methodology for the isolation of target cell populations from biological suspensions, covering a wide spectrum of applications from diagnosis and therapy in biomedicine to environmental applications or fundamental research in biology. This Special Issue aims to create a forum of discussion to share advances and address current challenges in magnetic cell separation. For more details, check out this flyer.
The deadline for your manuscript submission will be November 30, 2020.
May 01, 2020
 The European Magnetism Society just sent out a newsletter with a list of cancelled magnetism-related meetings. Check it out here:
The European Magnetism Society just sent out a newsletter with a list of cancelled magnetism-related meetings. Check it out here:
http://magnetism.eu/news/144/38-news.htm
Also, while the Covid-19 situation is still dire, the curve seems flattened in many countries, and governments are carefully starting to reopen schools etc. This also means that more people are looking for jobs again.
In the magnetism area, EMA also updated their lists of available jobs. Please check it out too:
http://magnetism.eu/5-job-market.htm
And if you are an employer who is looking for a new employee, please advertise at EMA, or also send us an e-mail so we can advertise on magneticmicrosphere.com.
April 20, 2020
 In the light of major conference cancellations, the magnetic particle imaging community has decided to maintain our fast progress in molecular imaging and support the trainees that are hard at work producing groundbreaking data.
In the light of major conference cancellations, the magnetic particle imaging community has decided to maintain our fast progress in molecular imaging and support the trainees that are hard at work producing groundbreaking data.
MPI is inviting researchers in the MPI field to participate in the Magnetic Particle Imaging Rising Stars 2020 Molecular Imaging eSymposia, taking place on April 21st and 22nd at 9 AM PT / 12 PM ET.
If you do some cool MPI research and would like to give a short 10 minute presentation, then please submit an abstract online at this address.
Abstract Deadline: April 3rd, 2020. For any questions, please contact Jeff Gaudet PhD.
April 08, 2020
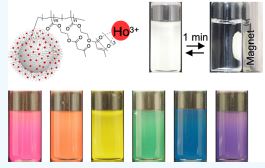 Michinari Kohri et al. just published a very cool paper in ACS Applied Polymer Materials. They demonstrated the production of colorless and full-color magnetic nanoparticles based on holmium (Ho)-doped polymers, which could not be achieved with conventional dark brown iron oxide magnetic nanoparticles. The coordination of Ho, a lanthanide with low colorability and a strong magnetic moment, with a poly(2-acetoacetoxy ethyl methacrylate) brush built on the surface of submicron-sized silica particles allowed for the formation of colorless magnetic nanoparticles. Additionally, bright and full-color magnetic nanoparticles were obtained by mixing different colored magnetic nanoparticles that were prepared by copolymerization of 2-acetoacetoxy ethyl methacrylate and dye monomers. Various colors, including transparency, were demonstrated by means of the present method, which determines the presence or absence of magnetism by Ho doping.
Michinari Kohri et al. just published a very cool paper in ACS Applied Polymer Materials. They demonstrated the production of colorless and full-color magnetic nanoparticles based on holmium (Ho)-doped polymers, which could not be achieved with conventional dark brown iron oxide magnetic nanoparticles. The coordination of Ho, a lanthanide with low colorability and a strong magnetic moment, with a poly(2-acetoacetoxy ethyl methacrylate) brush built on the surface of submicron-sized silica particles allowed for the formation of colorless magnetic nanoparticles. Additionally, bright and full-color magnetic nanoparticles were obtained by mixing different colored magnetic nanoparticles that were prepared by copolymerization of 2-acetoacetoxy ethyl methacrylate and dye monomers. Various colors, including transparency, were demonstrated by means of the present method, which determines the presence or absence of magnetism by Ho doping.
The bright and magnetically controllable colored nanoparticles presented herein may have a significant impact on practical substances and applications, such as ink and biomedical and device applications.
Check out the article here.

Beautiful ferrofluid, with a curious and striking 'peak-on-a-peak' effect. Submitted by Quentin Pankhurst.
Search this site with the power of
Last Modified: February 22, 2024 -
Magneticmicrosphere.com © 2024


 Researchers at Empa, the Swiss Federal Laboratories for Materials Science and Technology near Zurich have developed an innovative method to precisely produce different alloy compositions in a single component, using a 3D laser printer. Until recently, 3D printing has presented unique challenges to metal processing, as temperatures nearly instantaneously exceed 2,500 degrees Celsius in the melting zone, which can alter the properties of alloys as some components evaporate. However, a team at Empa, led by Aryan Arabi-Hashemi and Christian Leinenbach, discovered that by varying the power and duration of the laser beam, they could modify the alloys with micrometer precision, thereby “producing new materials with completely new functionalities.” Prospective applications include manufacturing “smart” shape memory alloys and designing more efficient electric motors.
Researchers at Empa, the Swiss Federal Laboratories for Materials Science and Technology near Zurich have developed an innovative method to precisely produce different alloy compositions in a single component, using a 3D laser printer. Until recently, 3D printing has presented unique challenges to metal processing, as temperatures nearly instantaneously exceed 2,500 degrees Celsius in the melting zone, which can alter the properties of alloys as some components evaporate. However, a team at Empa, led by Aryan Arabi-Hashemi and Christian Leinenbach, discovered that by varying the power and duration of the laser beam, they could modify the alloys with micrometer precision, thereby “producing new materials with completely new functionalities.” Prospective applications include manufacturing “smart” shape memory alloys and designing more efficient electric motors.
.JPG)





