
Guidelines for Dynamic Light Scattering Measurements and Analysis
May 26, 2014The company Nanocomposix published a very good introduction into particle size measurement with dynamic light scattering (DLS) measurement. After analyzing thousands of nanoparticle samples they have assembled a set of guidelines that can be used to determine how to maximize the quality of your DLS data and, more importantly, how to interpret your results. Check it out here. And if you have more questions, contact them at info@nanocomposix.com or (858) 565-4227 x 2.
World's Smallest Medical Robot
April 18, 2014
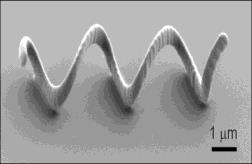 The smallest robot worldwide is only a few micrometer long and has been created at ETH Zurich. The robot designed to be steered through the human body forces high design chal-lenges on the researchers in terms of control and power supply. It is inspired by the flagella of the E. coli bacteria, which allows highly efficient locomotion. The metallic replica of the flagella is set in rotation by an external magnetic field, whose direction and intensity can be regulated and adjusted according to the direction the robot should swim. The ETH spin-off Aeon Scientific is currently working on the marketing of the new control technology.
The smallest robot worldwide is only a few micrometer long and has been created at ETH Zurich. The robot designed to be steered through the human body forces high design chal-lenges on the researchers in terms of control and power supply. It is inspired by the flagella of the E. coli bacteria, which allows highly efficient locomotion. The metallic replica of the flagella is set in rotation by an external magnetic field, whose direction and intensity can be regulated and adjusted according to the direction the robot should swim. The ETH spin-off Aeon Scientific is currently working on the marketing of the new control technology.
Hydrogel Micro-Robots Developed for Drug Delivery
April 18, 2014The toxicity of many drugs creates high risk of destroying healthy cells when attempting to treat disease. Scientists at ETH Zurich’s Institute of Robotics and Intelligent Systems (IRIS) have developed a tiny robot about a half-millimeter in diameter that may provide a solution. The robot comprises a star-shaped, bi-layer, soft hydrogel shell that, when closed, forms a sphere and holds tiny magnetic beads inside. When the robot reaches the desired loca-tion, laser irradiation causes the hydrogel to change shape and the capsule to open, delivering the drug. The hy-drogel robot has been tested only in-vitro, but the scientists believe they can miniaturize it enough to test it in ani-mals in the future.
Biosynthesis of Magnetic Nanostructures in a Foreign Organism by Transfer of Bacterial Magnetosome Gene Clusters
April 17, 2014
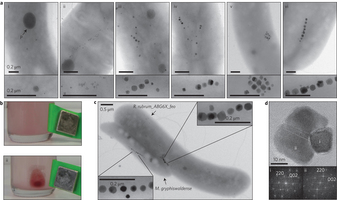 In nature, magnetosomes—membrane-bound magnetic nanocrystals with unprecedented magnetic properties—can be biomineralized by magnetotactic bacteria, e.g., Magnetospirillum gryphiswaldense . However, these microbes are difficult to handle. Isabel Kolinko, Youming Zhang, Dirk Schüler et al. now were able to transfer a minimum set of genes for the biosynthetic pathway of biomineralization of magnetic nanoparticles from these fastidious microorganisms to Rhodospirillum rubrum, a gram negative proteobacterium. Biomineralization of highly ordered magnetic nanostructures was then possible and might in the future allow for the sustainable production of tailored magnetic nanostructures in biotechnologically relevant hosts. This represents a step towards the endogenous magnetization of various organisms by synthetic biology.
In nature, magnetosomes—membrane-bound magnetic nanocrystals with unprecedented magnetic properties—can be biomineralized by magnetotactic bacteria, e.g., Magnetospirillum gryphiswaldense . However, these microbes are difficult to handle. Isabel Kolinko, Youming Zhang, Dirk Schüler et al. now were able to transfer a minimum set of genes for the biosynthetic pathway of biomineralization of magnetic nanoparticles from these fastidious microorganisms to Rhodospirillum rubrum, a gram negative proteobacterium. Biomineralization of highly ordered magnetic nanostructures was then possible and might in the future allow for the sustainable production of tailored magnetic nanostructures in biotechnologically relevant hosts. This represents a step towards the endogenous magnetization of various organisms by synthetic biology.
Check out the details in this interesting Nature Nanotechnology 9, 193-197 (2014) paper. It includes an editorial.
Visualizing the Tree-Rings of Magnetic Fields
March 23, 2014
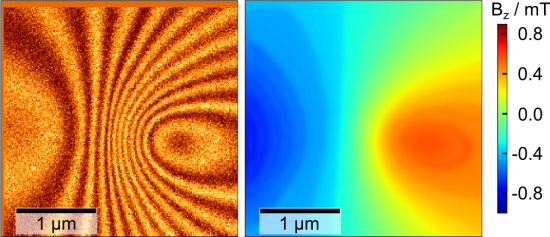 Microscopes able to image magnetic fields are an increasingly important tool, not only for material physics but also for industry where they are used to check the magnetic field of hard disk write heads. The NV center in diamond – an atomic-size color defect – is a promising candidate for the next generation of sensor heads for magnetic field microscopes. It can sense magnetic fields with high (nT) sensitivity and, more importantly, sub-nm resolution. This latter property is an order of magnitude better than existing techniques such as magnetic force microscopy.
Microscopes able to image magnetic fields are an increasingly important tool, not only for material physics but also for industry where they are used to check the magnetic field of hard disk write heads. The NV center in diamond – an atomic-size color defect – is a promising candidate for the next generation of sensor heads for magnetic field microscopes. It can sense magnetic fields with high (nT) sensitivity and, more importantly, sub-nm resolution. This latter property is an order of magnitude better than existing techniques such as magnetic force microscopy.
Imaging of magnetic fields with NV centers has been demonstrated before, but has so far required sophisticated and error-prone control techniques (e.g. lock-in detection). Prof. Wachtrup in Stuttgart and his team now managed to radically simplify this technique with the help of a computer-optimized spectroscopy protocol based on a technique known as optimal control. His protocol switches off fluorescence of a scanning color center whenever the magnetic field reaches one of several magic values. Scanning through a spatially varying magnetic field, fluorescence of the center simultaneously images multiple contour lines of the magnetic field – lines where the field takes on one of the magic values. This pattern is visually and conceptually analogous to tree lines in biology, which – mathematically phrased – reveal all parts of a piece of wood that were grown by the tree at a particular instant in time. Technically, this contour line pattern can be used to quantitatively reconstruct a map of the full magnetic field, just as tree-rings can reveal the growth history of a tree.
The inventors demonstrated their technique by imaging a MFM-tip as an example of a magnetic nanostructure, gaining previously unknown insights into its magnetic field structure. It was then used to check different models of the MFM-tip's magnetic field for validity.
For more information, check here.
Dynamic Magnetic Fields Remote Control Apoptosis Via Nanoparticle Rotation
March 08, 2014
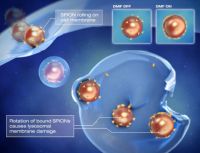 Enming Zhang, Erik Renström, Martin Koch et al. examined whether the rotational nanoparticle movement could be used for remote induction of cell death by injuring lysosomal membrane structures - without magnetic hyperthermia. They further hypothesized that the shear forces created by the generation of oscillatory torques (incomplete rotation) of SPIONs bound to lysosomal membranes would cause membrane permeabilization, lead to extravasation of lysosomal contents into the cytoplasm, and induce apoptosis. To examine this experimentally, they covalently conjugated SPIONs with antibodies targeting the lysosomal protein marker LAMP1 (LAMP1-SPION).
Enming Zhang, Erik Renström, Martin Koch et al. examined whether the rotational nanoparticle movement could be used for remote induction of cell death by injuring lysosomal membrane structures - without magnetic hyperthermia. They further hypothesized that the shear forces created by the generation of oscillatory torques (incomplete rotation) of SPIONs bound to lysosomal membranes would cause membrane permeabilization, lead to extravasation of lysosomal contents into the cytoplasm, and induce apoptosis. To examine this experimentally, they covalently conjugated SPIONs with antibodies targeting the lysosomal protein marker LAMP1 (LAMP1-SPION).
Remote activation of slow rotation of LAMP1-SPIONs significantly improved the efficacy of cellular internalization of the nanoparticles. Furthermore, the magnetic nanoparticles induced both early and late apoptosis remotely. Dynamic magnetic field generation, which the authors termed DMF, might thus be an important platform technology for novel biomedical applications.
Check the article and details for yourself here.
Nanoparticles and magnetism might enhance immune response to viruses and cancer
February 28, 2014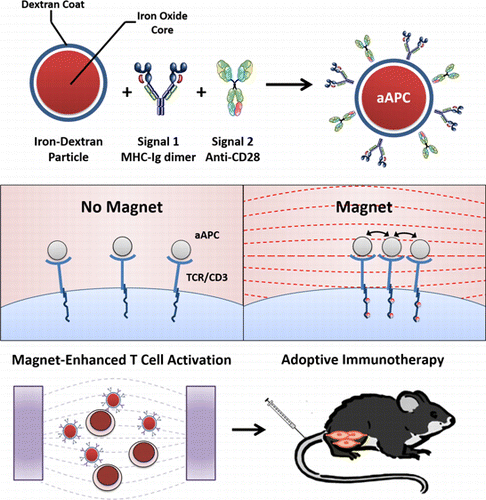 Johns Hopkins University researchers have used nanoparticles and magnets to enhance the endogenous immune response to viruses and cancers. When the approach was tested on mice with skin tumors, six of eight that were treated survived longer than four weeks with no signs of tumor growth, while none of the untreated mice survived. Details of the study appear in the journal ACS Nano.
Johns Hopkins University researchers have used nanoparticles and magnets to enhance the endogenous immune response to viruses and cancers. When the approach was tested on mice with skin tumors, six of eight that were treated survived longer than four weeks with no signs of tumor growth, while none of the untreated mice survived. Details of the study appear in the journal ACS Nano.“Size was key to this experiment,” says Jonathan Schneck, M.D., Ph.D., a professor of pathology, medicine and oncology at the Johns Hopkins University School of Medicine’s Institute for Cell Engineering. “By using small enough particles, we could, for the first time, see a key difference in cancer-fighting cells, and we harnessed that knowledge to enhance the immune attack on cancer.”
Dr. Schneck’s team has pioneered the development of artificial white blood cells, so-called artificial antigen-presenting cells (aAPCs), which show promise in training animals’ immune systems to fight diseases such as cancer. To do that, the aAPCs must interact with naive T cells that are already present in the body, awaiting instructions about which specific invader they will battle. The aAPCs bind to specialized receptors on the T cells’ surfaces and present them with distinctive proteins called antigens. This process activates the T cells, programming them to battle a specific threat such as a virus, bacteria or tumor, as well as to make more T cells.
Superconductivity Switched on by Magnetic Field
February 16, 2014
 Strong magnetic fields typically interfere with superconductivity. However, researchers at the Paul Scherrer Institute have discovered a material, CeCoIn5, where a magnetic field creates superconductivity. The superconducting state is in addition to, and simultaneous with, a first superconducting state that appears at low temperatures. Furthermore, an addi-tional antiferromagnetic order was observed, and it was discovered with the SINQ neutron source. This discovery ultimately demonstrates the direct control of quantum states, an im-portant discovery for future quantum computers.
Strong magnetic fields typically interfere with superconductivity. However, researchers at the Paul Scherrer Institute have discovered a material, CeCoIn5, where a magnetic field creates superconductivity. The superconducting state is in addition to, and simultaneous with, a first superconducting state that appears at low temperatures. Furthermore, an addi-tional antiferromagnetic order was observed, and it was discovered with the SINQ neutron source. This discovery ultimately demonstrates the direct control of quantum states, an im-portant discovery for future quantum computers.
http://www.psi.ch/media/superconductivity-switched-on-by-magnetic-field
For more information, check out our Archives.
September 2017

Search this site with the power of
